维权声明:本文为Insp_4125fa38原创作品,本作者与仪器信息网是该作品合法使用者,该作品暂不对外授权转载。其他任何网站、组织、单位或个人等将该作品在本站以外的任何媒体任何形式出现均属侵权违法行为,我们将追究法律责任。
桔梗化学成分的提取分离
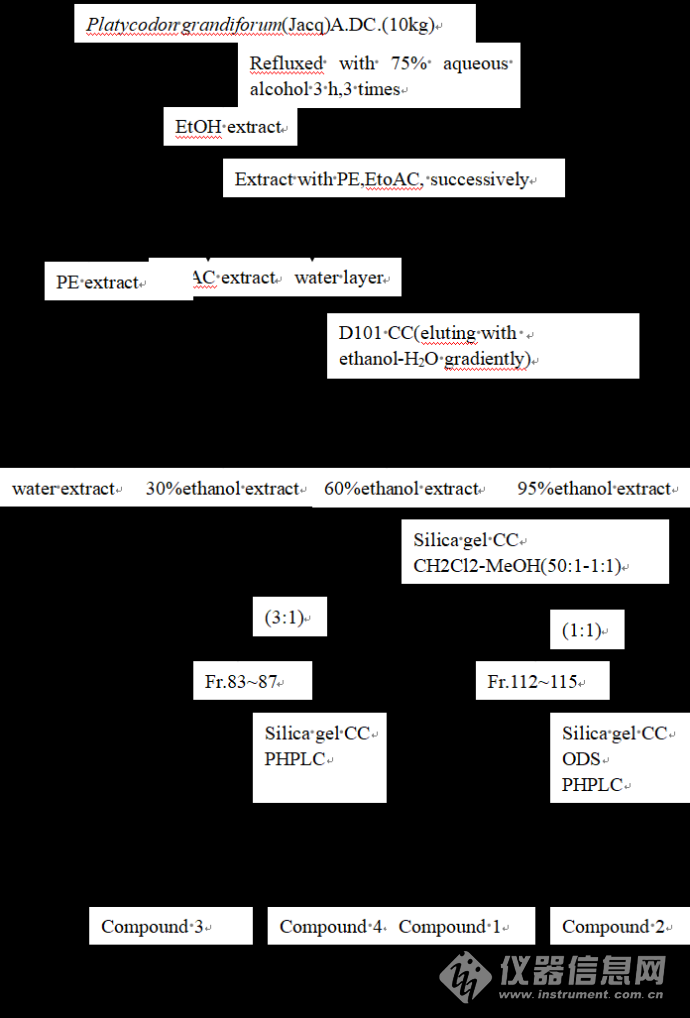
1 桔梗化学成分提取分离流程桔梗干燥根药材(10 kg),置于大型中药提取器中,以6倍量75%的乙醇浸泡过夜,加热回流提取3次,每次3 h,合并三次提取液,减压回收乙醇。将浓缩液用等体积的石油醚、乙酸乙酯分别萃取3次,剩余水溶液浓缩约3 L,经D101大孔吸附树脂,分别用水,30%乙醇水,60%乙醇水和95%乙醇水依次洗脱,得60%乙醇水洗脱部分浓缩后得浸膏90 g。60 %乙醇洗脱部分经(200-300目)柱色谱,以二氯甲烷-甲醇梯度洗脱(50:1;25:1;12:1;6:1;3:1;1:1)。将二氯甲烷-甲醇(1:1)馏分再经硅胶柱层析、反相ODS柱色谱,反相半制备高效液相色谱最终分离得到白色粉末状Compound 1,Compound 2;将二氯甲烷-甲醇(3:1)馏分再经硅胶柱层析、反相制备高效液相色谱,反相半制备高效液相色谱最终分离得到白色粉末状Compound 3和Compound 4。其具体分离流程如图1所示:![]()
图1 提取分离流程图
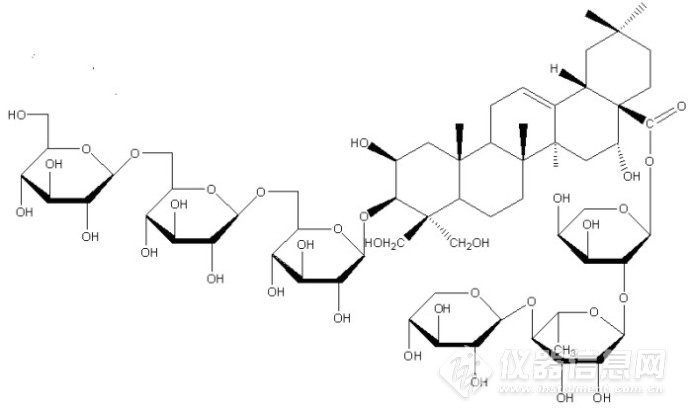
[img=,8,]file:///C:/Users/zhang/AppData/Local/Temp/ksohtml9420/wps11.png[/img][img=,8,]file:///C:/Users/zhang/AppData/Local/Temp/ksohtml9420/wps12.png[/img][img=,8,]file:///C:/Users/zhang/AppData/Local/Temp/ksohtml9420/wps13.png[/img][img=,8,]file:///C:/Users/zhang/AppData/Local/Temp/ksohtml9420/wps14.png[/img]2 化合物结构鉴定Compound 1:白色粉末(甲醇)。1H-NMR(300MHz ,pyridine-d5) δ:1.00(3H,s,H-29), 1.08(3H,s,H-30), 1.12(3H,s,H-26), 1.39(3H,s,H-27), 1.68(3H,s,H-25),1.68(3H,br.s,CH3-Rha)及不饱和双键氢δ5.63(1H,br.s,12-H)。13C-NMR(150MHz, pyridine-d5)δ:45.3(C-1), 68.7(C-2), 88.7(C-3), 48.2(C-4), 47.6(C-5), 19.4(C-6), 33.3(C-7), 40.6(C-8), 45.3(C-9), 38.0(C-10), 24.1(C-11), 123.3(C-12), 144.4(C-13), 42.5(C-14), 36.1(C-15), 74.0(C-16), 49.7(C-17), 41.6(C-18), 47.2(C-19), 31.0(C-20), 36.1(C-21), 32.2(C-22), 63.2(C-23), 67.5(C-24), 19.1(C-25), 17.7(C-26), 27.1(C-27), 176.0(C-28), 33.6(C-29), 24.8(C-30)。13C-NMR data of sugars:Glucose(inner): 106.1(C-1), 74.9(C-2), 78.4(C-3),72.4(C-4),76.5(C-5),70.4(C-6); Glucose(center): 105.0(C-1), 75.3(C-2),78.5(C-3), 71.3(C-4), 77.2(C-5),70.2(C-6); Glucose(terminal):105.6(C-1), 75.4(C-2),78.7(C-3),70.9(C-4),77.7(C-5),62.7(C-6); Arabinose: 93.6(C-1), 75.3(C-2), 71.6(C-3), 66.3(C-4), 63.3(C-5); Rhamnose: 101.2(C-1), 72.0(C-2), 72.7(C-3), 83.6(C-4), 68.7(C-5), 18.4(C-6); Xylose: 106.9(C-1), 76.1(C-2), 78.6(C-3), 71.1(C-4), 67.5(C-5)。该化合物数据与文献报道基本一致]。故鉴定化合物1为去芹糖桔梗皂苷E,结构式如图2所示。![]()
图2 化合物1结构式
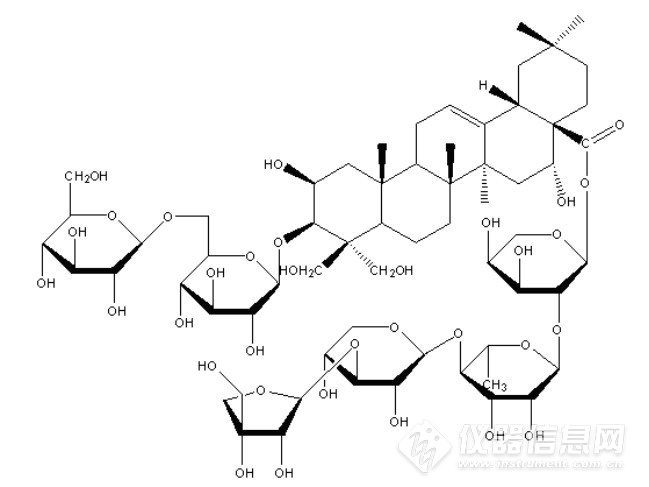
Compound 2:白色粉末(甲醇)。1H-NMR(300MHz,pyridine-d5)δ:1.00(3H,s,H-29), 1.11(3H,s,H-30), 1.08(3H,s,H-26), 1.36(3H,s,H-27), 1.69(3H,s,H-25),1.69(3H,br.s,CH3-Rha)及不饱和双键氢δ5.80(1H,br.s,12-H)。13C-NMR(150MHz ,pyridine-d5)δ:45.3(C-1),68.8(C-2),88.4(C-3),48.2(C-4),47.6(C-5),19.5(C-6),33.5(C-7),40.6(C-8),45.0(C-9),38.0(C-10),24.1(C-11),123.3(C-12),144.4(C-13),42.5(C-14),36.1(C-15),73.9(C-16),49.8(C-17),41.7(C-18),47.2(C-19),31.0(C-20),36.1(C-21),32.2(C-22),63.6(C-23),67.0(C-24),19.2(C-25),17.7(C-26),27.1(C-27),176.1(C-28),33.3(C-29),24.8(C-30)。13C-NMR data of sugars:Glucose(inner):106.1(C-1),74.9(C-2),78.4(C-3),72.4(C-4),76.6(C-5),70.6(C-6); Glucose(center): 105.0(C-1), 75.3(C-2),78.5(C-3), 71.3(C-4), 77.2(C-5),70.2(C-6); Glucose(terminal):105.7(C-1), 75.2(C-2),78.7(C-3),70.9(C-4),77.7(C-5),62.7(C-6); Arabinose: 93.7(C-1), 75.5(C-2), 71.6(C-3), 66.5(C-4), 63.1(C-5); Rhamnose: 101.3(C-1), 71.9(C-2), 72.8(C-3), 83.9(C-4), 68.6(C-5), 18.4(C-6); Xylose: 106.8(C-1), 75.8(C-2), 84.9(C-3), 69.5(C-4), 67.0(C-5); Apiose:111.3(C-1), 77.9(C-2), 80.6(C-3), 75.3(C-4), 65.4(C-5)。该化合物数据与文献报道基本一致,故鉴定化合物2为桔梗皂苷E,结构式如图3所示。![]()
图3 化合物2结构式
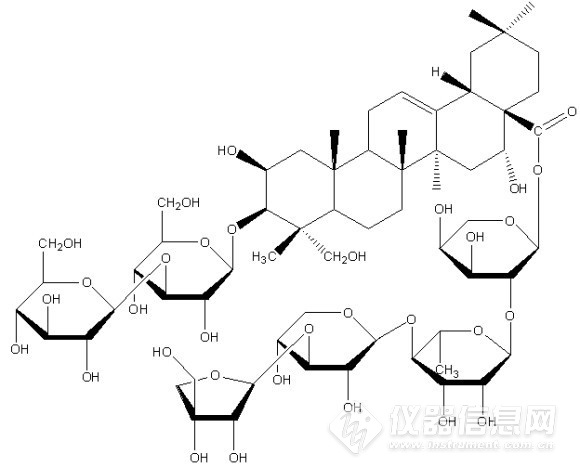
Compound 3:白色粉末(甲醇)。1H-NMR(300MHz,pyridine-d5)δ:0.99(3H,s,H-29), 1.07(3H,s,H-30), 1.11(3H,s,H-26), 1.35(3H,s,H-27), 1.68(3H,s,H-25),1.68(3H,br.s,CH3-Rha)及不饱和双键氢δ5.63(1H,br.s,12-H)。13C-NMR(75MHz,pyridine-d5) δ:45.0(C-1), 68.2(C-2), 88.1(C-3), 47.2(C-4), 47.9(C-5), 19.2(C-6), 33.2(C-7), 40.2(C-8), 44.5(C-9), 37.7(C-10), 24.1(C-11), 123.3(C-12), 144.0(C-13), 42.1(C-14), 35.7(C-15), 73.6(C-16), 49.4(C-17), 41.3(C-18), 46.8(C-19), 30.6(C-20), 35.7(C-21), 31.8(C-22), 63.3(C-23), 67.1(C-24), 18.8(C-25), 17.3(C-26), 26.7(C-27), 175.7(C-28), 32.9(C-29), 24.4(C-30)。13C-NMR data of sugars:Glucose(inner):105.8(C-1),74.5(C-2),78.3(C-3),71.8(C-4),76.2(C-5),70.2(C-6); Glucose(terminal): 104.7(C-1),75.2(C-2),78.5(C-3),69.1(C-4),77.5(C-5),62.3(C-6); Arabinose: 93.3(C-1), 76.2(C-2), 66.2(C-3), 70.2(C-4), 62.7(C-5);Rhamnose: 100.9(C-1), 71.1(C-2), 72.4(C-3), 83.6(C-4), 68.4(C-5), 18.1(C-6);Xylose: 106.4(C-1), 75.2(C-2), 84.4(C-3), 70.2(C-4), 66.6(C-5); Apiose:110.9(C-1), 77.5(C-2), 80.2(C-3), 74.9(C-4), 65.0(C-5)。该化合物数据与文献报道基本一致[。故鉴定化合物3为桔梗皂苷D3,结构式如图4所示。![]()
图4 化合物3结构式
Compound 4:白色粉末(甲醇)。1H-NMR(600MHz,pyridine-d5)δ:0.98(3H,s,H-29), 1.13(3H,s,H-26), 1.14(3H,s,H-30), 1.33(3H,s,H-25), 1.56(3H,s,H-24),1.76(3H,s,H-27), 1.70(3H,br.s,CH3-Rha)及不饱和双键氢δ5.72(1H,br.s, H-12)。13C-NMR(150MHz,pyridine-d5) δ:44.4(C-1), 69.7(C-2), 83.7(C-3), 42.9(C-4), 47.7(C-5), 18.1(C-6), 33.3(C-7), 40.2(C-8), 47.8(C-9), 37.0(C-10), 24.1(C-11), 123.2(C-12), 144.4(C-13), 42.3(C-14), 36.0(C-15), 74.1(C-16), 49.6(C-17), 41.3(C-18), 47.1(C-19), 32.2(C-20), 36.2(C-21), 31.0(C-22), 65.2(C-23), 15.1(C-24), 17.7(C-25), 17.4(C-26), 27.3(C-27), 176.0(C-28), 33.3(C-29), 24.8(C-30)。13C-NMR data of sugars:Glucose(inner):105.9(C-1),74.2(C-2),88.7(C-3),72.0(C-4),77.8(C-5),70.8(C-6); Glucose(terminal): 105.4(C-1),75.4(C-2),78.8(C-3),69.4(C-4),78.3(C-5),62.3(C-6); Arabinose: 93.5(C-1), 75.6(C-2), 66.0(C-3), 69.9(C-4), 62.6(C-5);Rhamnose: 101.2(C-1), 71.7(C-2), 72.7(C-3), 83.0(C-4), 68.6(C-5), 18.4(C-6);Xylose: 106.7(C-1), 75.2(C-2), 84.8(C-3), 69.1(C-4), 67.0(C-5); Apiose:111.3(C-1), 77.9(C-2), 80.5(C-3), 75.3(C-4), 65.4(C-5)。该化合物数据与文献报道基本一致。故鉴定化合物4为远志皂苷D2,结构式如图2所示。![]()
图5 化合物4结构式
3 小结 采用75 %乙醇回流提取,再分别用石油醚、乙酸乙酯进行萃取,剩余水溶液浓缩后通过D101大孔吸附树脂,对60 %乙醇洗脱液进行提取分离。通过采用反复硅胶柱色谱法、ODS柱色谱和制备以及半制备高效液相色谱法等分离纯化手段,共分离纯化得到4个化合物,均为桔梗皂苷类化合物,鉴定为:去芹糖桔梗皂苷E(Deapio-platycoside E)、桔梗皂苷E(Platycoside E)、桔梗皂苷D3(Platycodin D3)和远志皂苷D2(Polygalacin D2)。 桔梗中主要药理活性成分为桔梗皂苷,含量较大,利用传统的萃取技术来纯化大量皂苷震摇剧烈容易产生乳化现象,不利于纯化,因此先使用大孔吸附树脂对皂苷成分进行前期纯化,有助于富集大量的皂苷进行后续的单体化合物的分离。 桔梗皂苷为齐墩果酸型五环三萜类皂苷,一般母核连有3~7个糖基,极性较大,而且结构相近较难分离,采用硅胶柱层析等正相分离手段无法较好的分离,必须较多的借助反复ODS柱色谱,制备高效液相色谱等反相分离技术得以分离得到较纯的单体化合物。