8. Environmental Contamination
During the course of a sample preparation, contamination can occur from the environment, the reagents, the apparatus, and even the analyst. In many cases the blank determines the lower limit of detection. The next several parts of this guide deal with contamination.
Overview
Environmental contamination is caused by particulate and/or gaseous matter in the air. It has been reported that air in an analytical laboratory can contain up to 200 µg/m3 of particulate matter containing Ca, Si, Fe, Na, Mg, K, Tl, Cu, Mn, and lesser amounts of other elements1. In addition, this reference states that normal rural area airborne particle counts have been reported to be 1,400,000/m3 for particles greater than 0.5 µm and that normal metropolitan area particle counts have been reported to be 53,000,000/m3 for particles greater than 0.5 µm.
Reducing Environmental Contamination:
Use the chemical blank as the performance criteria. Dealing with environmental contamination is expensive. Therefore, reduce the blank to the lowest possible value by eliminating contamination from the sample container, the chemist, and sample preparation apparatus. Achieve this by doing the following:
Separate the sample preparation and handling area from all other areas.
Eliminate as many foreign objects as possible from the preparation area (i.e. - Motorized objects such as stirring plates).
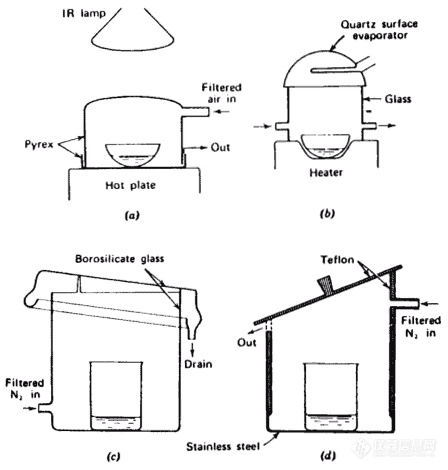
Use all-plastic hoods.
Keep the sample out of the hood area as much as possible.
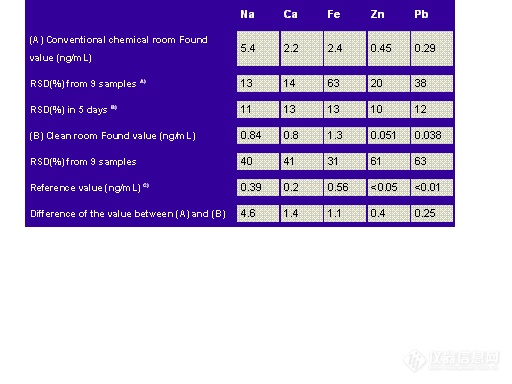
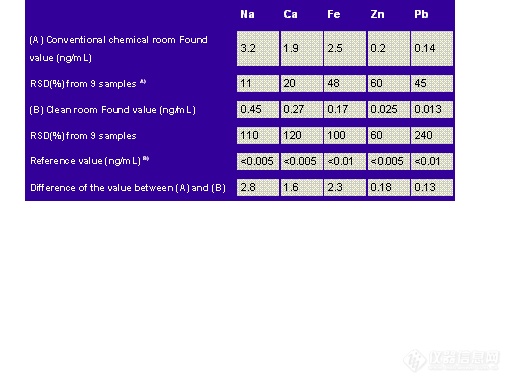
The effect of the laboratory atmosphere on lead blank levels is shown in Table 8.1.
Table 8.1: Effect of Laboratory Atmosphere on Pb Blank Levels
![]()
Hoods, which enhance atmospheric exposure and hence contamination, are necessary for the safety of the analyst. Atmospheric contamination has been reduced significantly through the use of "clean rooms" that use HEPA filtered air. HEPA (High Efficiency Particulate Air) filters are 99.99 % efficient in removing particulates down to 0.3 µm.