Common Twin Laws 1.
Triclinic System - The feldspar minerals plagioclase and microcline are the most common triclinic minerals that show twinning. Two common twin laws are observed in these feldspars.
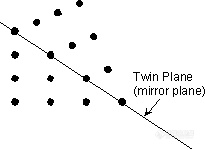
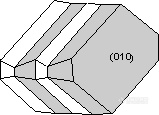
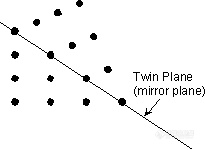
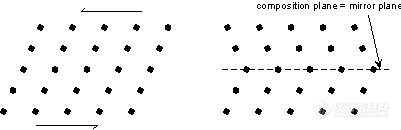
。Albite Law - As described above, plagioclase (NaAlSi3O8 - CaAl2Si2O8) very commonly shows albite polysynthetic twinning. The twin law - {010} indicates that the twining occurs perpendicular to the b crystallographic axis. Albite twinning is so common in plagioclase, that it's presence is a diagnostic property for identification of plagioclase.
![]()
。Pericline Law - The pericline law has [010] as the twin axis. As stated above, pericline twinning occurs as the result of monoclinic orthoclase or sanidine transforming to microcline (all have the same chemical formula - KAlSi3O8). Pericline twinning usually occurs in combination with albite twinning in microcline, but is only observable with the polarizing microscope. The combination of pericline and albite twinning produce a cross-hatched pattern, called tartan twinning, as discussed above, that easily distinguishes microcline from the other feldspars under the microscope.
2.
Monoclinic System - The most common twins in the monoclinic system occur on the planes {100} and {001}. The feldspars - orthoclase and sanidine - are the most commonly twinned minerals in the monoclinic system. Both contact twins and penetration twins occur, and both types result from accidents during growth.
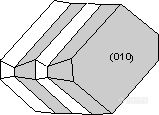
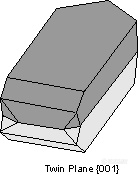
。Manebach Law - {001} - forms a contact twin commonly observed in the mineral orthoclase. This twinning is very diagnostic of orthoclase when it occurs.
![]()
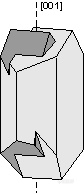
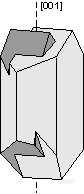
。Carlsbad Law - [001] - forms a penetration twin in the mineral orthoclase. Crystals twinned under the Carlsbad Law show two intergrown crystals, one rotated 180o from the other about the [001] axis. Carlsbad twinning is the most common type of twinning in orthoclase, and is thus very diagnostic of orthoclase when it occurs.
![]()
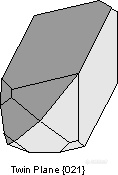
。Braveno Law - {021} - forms a contact twin in the mineral orthoclase.
![]()
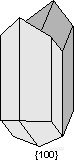
。Swallow Tail Twins - {100}- are commonly observed in the mineral gypsum (CaSO4.2H2O).
![]()
3.
Orthorhombic System - Orthorhombic crystals commonly twin on planes parallel to a prism face. The most common is a {110} twin that results in many orthorhombic minerals having cyclical twins.
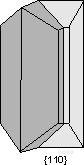
。{110} Cyclical Twins - The mineral aragonite (CaCO3) , chrysoberyl (BeAl2O4), and cerrusite (PbCO3) commonly develop twinning on {110}. This results in a cyclical twin which gives these minerals a pseudo-hexagonal appearance.
![]()
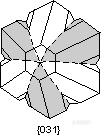
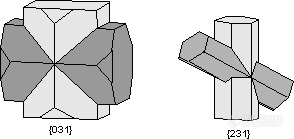
。Staurolite Law - The mineral staurolite is really monoclinic, but it has a ß angle very close to 90o so it has the appearance of an orthorhombic mineral. Two types of interpenetration twins occur in staurolite the {031} twins from a right-angled cross and the {231} twins form a cross at about 60o.
![]()
4.
Tetragonal System - Twinning in the tetragonal system usually occurs on {011} forming cyclical contact twins. The minerals rutile (TiO2) and cassiterite (SnO2) commonly show this type of twinning.
![]()
5.
Hexagonal System - The minerals calcite (CaCO3) and quartz (SiO2) are the most common hexagonal minerals and both show the types of twinning common in hexagonal minerals.
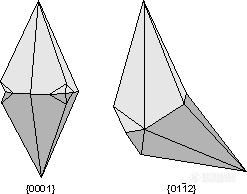
。Calcite Twins - The two most common twin laws that are observed in calcite crystals are {0001} and the rhombohedron {01-12}. Both are contact twins, but the {01-12} twins can also occur as polysynthetic twins that result from deformation.
![]()
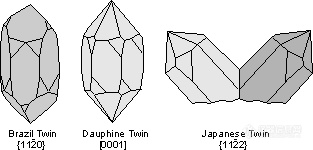
Quartz shows three other hexagonal twins.
。Brazil Law - {11-20} - is a penetration twin that results from transformation.
。Dauphiné Law - [0001] - is also a penetration twin that results from transformation.
。Japanese Law - {11-22} - is a contact twin that results from accidents during growth.
![]()
6.
Isometric System - Three types of twins are common in the isometric system.
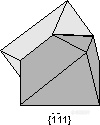
。Spinel Law - {-1-11} - is a twin plane, parallel to an octahedron. It occurs commonly in mineral spinel (MgAl2O4).
![]()
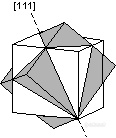
。[111] - The twin axis perpendicular to an octahedral face adds three fold rotational symmetry.
![]()
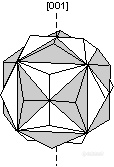
。Iron Cross [001] - The mineral pyrite (FeS2) often shows the iron cross made of the interpenetration of two pyritohedrons. Since this occurs in the class 2/m-3, with no 4-fold rotation axes, the [001] twin axis gives the mineral apparent 4-fold symmetry about 3 perpendicular axes.
![]()
转贴完

 ,问题是FCC的111面太特殊。这样看来,旋转孪晶对ABC的等价性没影响?!有实践经验的指教一下。哈
,问题是FCC的111面太特殊。这样看来,旋转孪晶对ABC的等价性没影响?!有实践经验的指教一下。哈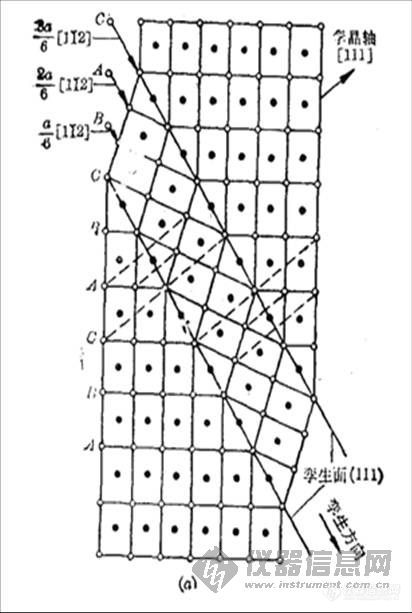
 twinninig in crystal
twinninig in crystal