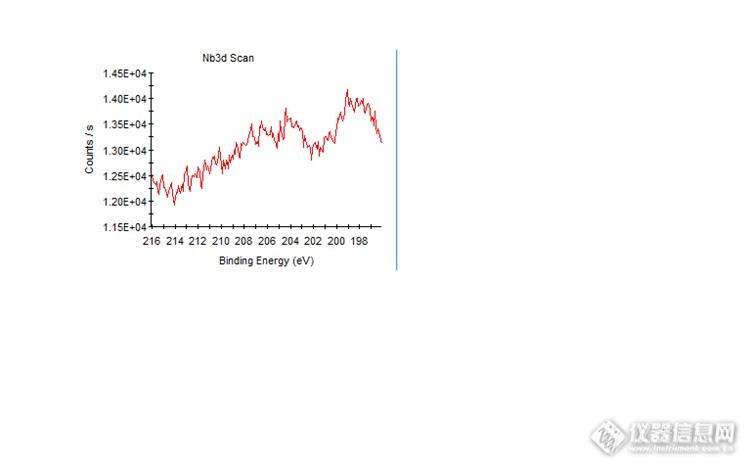
You cannot judge just by this narrow-spectrum. On the Nd3d peak there should be a double of 3d5 and 3d3.
If it is a metal Nd, Nd3d5 should be near 202eV BE and 3d3 at 205.2eV BE. But the peaks you have seems to be near 204eV indicating even Nd is existed it maybe in an Oxide form.
***There looks like a peak near 198eV BE too but it is almost no way for the Nd3d peak to shift to lower binding energy in Chemical form. Or unless you have some sample charging effect on your sample?
Going to next step if you want to "believe" the peak near 204eV is really Nd3d and looking at your spectrum this "peak" have an intensity of 0.05e+4 (peak to background). Then if you want to prove what you "believe" then you can look at Nd3p which is around 360eV BE. From standard spectra checking, if Nd is really presence, then Nd3p should have an intensity about half to Nd3d. So if you can see a peak at ~360eV BE and intensity is roughly like 0.025e+4 (peak to BG) then you can double confirm it is indeed the Nd being presence.
The peaks in your spectrum are really quite small, so it is indeed difficult to judge if such an element really presence or not. But with another small peaks in what so-called the same family peaks to come up, it can be a good proof for a presence of an element.
Hope you enjoy XPS~