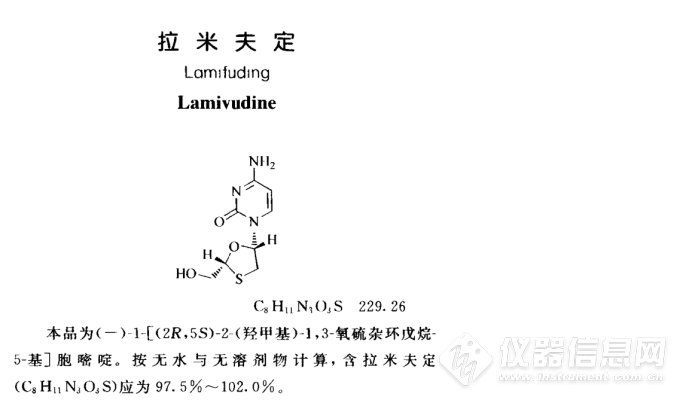
来自中国的自信,月旭Welchrom® C18测定拉米夫定有关物质
除非经大量试验比较市面主流厂家的色谱柱均不能达到要求,最好不要把色谱柱的品牌定下来。拉米夫定(Lamivudine)对乙型肝炎病毒和HIV有明显的抑制作用。口服吸收后,拉米夫定可在HIV感染细胞和正常细胞内代谢生成拉米夫定三磷酸盐,它是拉米夫定的活性形式。后者通过竞争抑制作用,终止DNA链的延长,从而抑制HIV和HBV的反转录酶和HBV聚合酶,阻止HIV和HBV的DNA合成和病毒复制。体外实验中与齐多夫定联合,对HIV病毒有协同作用。
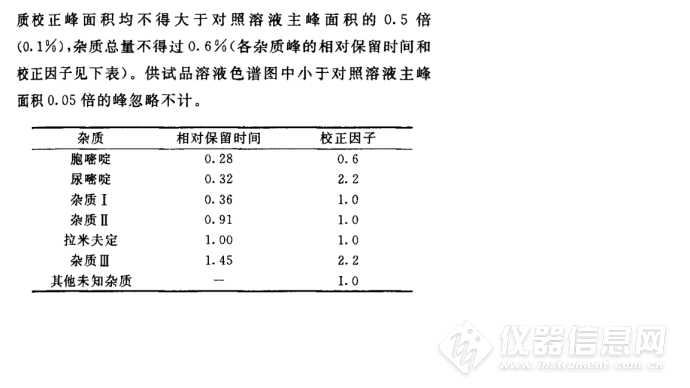
下面为中国药典2015年版收载的质量标准,其中提到的色谱柱,我们咨询了价格比较贵,我们就用手上的月旭色谱柱进行了探索研究。参照了EP8.0质量标准,进行了系统适用性试验,其中EP8.0质量标准有关物质项下为:
Related substances. Liquid chromatography (2.2.29).
Test solution. Dissolve 50.0 mg of the substance to be
examined in the mobile phase and dilute to 100.0 mL with
the mobile phase.
Reference solution (a). Dilute 1.0 mL of the test solution to
100.0 mL with the mobile phase. Dilute 1.0 mL of this solution
to 10.0 mL with the mobile phase.
Reference solution (b). Dissolve 5 mg of salicylic acid R in the
mobile phase and dilute to 100.0 mL with the mobile phase.
Dilute 1.0 mL of the solution to 100.0 mL with the mobile
phase.
Reference solution (c). Dissolve 50.0 mg of lamivudine CRS
in the mobile phase and dilute to 100.0 mL with the mobile
phase.
Reference solution (d). Dissolve 5 mg of cytosine R and 5 mg of
uracil R in the mobile phase and dilute to 100.0 mL with the
mobile phase. Dilute 2.0 mL of the solution to 10.0 mL with
the mobile phase.
Reference solution (e). Dissolve 5 mg of lamivudine for system
suitability 1 CRS (containing impurities A and B) in 2 mL of
the mobile phase. Add 1.0 mL of reference solution (d) and
dilute to 10.0 mL with the mobile phase.
Column:
- size: l = 0.25 m, Ø = 4.6 mm;
- stationary phase: base-deactivated octadecylsilyl silica gel for
chromatography R (5 μm);
- temperature: 35 °C.
Mobile phase: mix 5 volumes of methanol R and 95 volumes of
a 1.9 g/L solution of ammonium acetate R, previously adjusted
to pH 3.8 with glacial acetic acid R.
Flow rate: 1.0 mL/min.
Detection: spectrophotometer at 277 nm.
Injection: 10 μL.
Run time: 3 times the retention time of lamivudine.
Identification of impurities: use the chromatograms obtained
with reference solutions (b) and (e) to identify the peaks due
to impurities A, B, E, F and C.
Relative retention with reference to lamivudine (retention
time = about 9 min): impurity E = about 0.28;
impurity F = about 0.32; impurity A = about 0.36;
impurity B = about 0.91; impurity J = about 1.45;
impurity C = about 2.32.System suitability: reference solution (e):
- resolution: minimum 1.5 between the peaks due to
impurities F and A; minimum 1.5 between the peaks due
to impurity B and lamivudine.
Limits:
- correction factors: for the calculation of content,
multiply the peak areas of the following impurities by
the corresponding correction factor: impurity E = 0.6;
impurity F = 2.2; impurity J = 2.2;
- impurity A: not more than 3 times the area of the principal
peak in the chromatogram obtained with reference
solution (a) (0.3 per cent);
- impurity B: not more than twice the area of the principal
peak in the chromatogram obtained with reference
solution (a) (0.2 per cent);
- impurity C: not more than the area of the principal peak
in the chromatogram obtained with reference solution (b)
(0.1 per cent);
any other impurity: for each impurity, not more than the
area of the principal peak in the chromatogram obtained
with reference solution (a) (0.1 per cent);
- total: not more than 6 times the area of the principal peak
in the chromatogram obtained with reference solution (a)
(0.6 per cent);
- disregard limit: 0.5 times the area of the principal peak in
the chromatogram obtained with reference solution (a)
(0.05 per cent).
中文名拉米夫定外文名lamivudine
CAS号134678-17-4分子式C8H11N3O3S
![]()
![]()
![]()
![]()
![]()
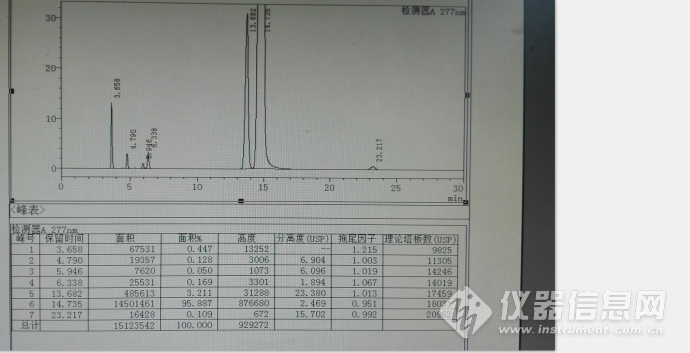
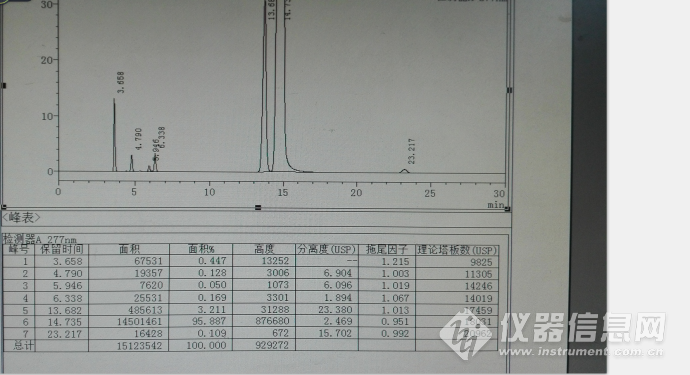
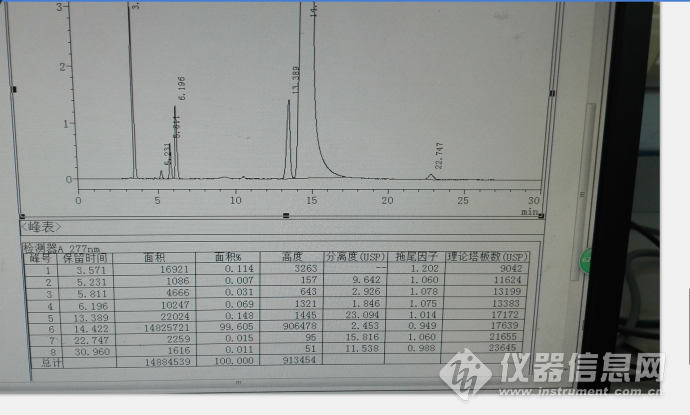
以上均为参照中国药典2015年收载的拉米夫定片质量的截图,然后进行系统适用性试验,进行流动相微调,其典型色谱图见下图:
![]()
初步统计色谱峰保留时间,其他色谱图参数很好,理论板数都超级好,分离度和拖尾因子也超级好!
| 相对保留时间(RRT) | 保留时间(min) |
| 0.25 | 3.658 |
| 0.33 | 4.79 |
| 0.40 | 5.946 |
| 0.43 | 6.338 |
| 0.93 | 13.682 |
| 1.00 | 14.735 |
| 1.58 | 23.217 |
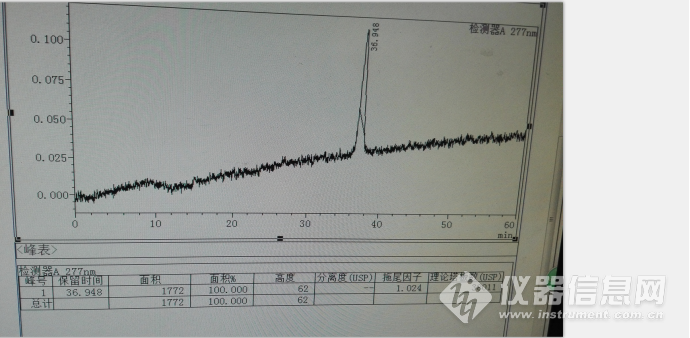
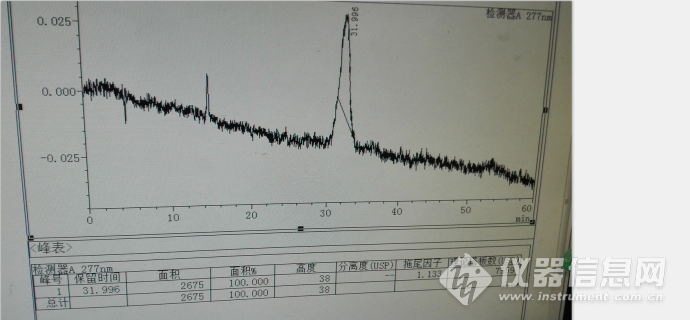
其中水杨酸的色谱峰如下:
![]()
另外最好控制柱温,最好是
液相自动控温装置,不要外接质量差的柱温箱,因为在升温的过程有±5℃的差异,会引起水杨酸色谱峰保留时间和峰型变化较大,使其用相对保留时间定位出现偏差,建议用水杨酸对照品同时定位,因为其价格便宜易得,其典型色谱图如下:
![]()
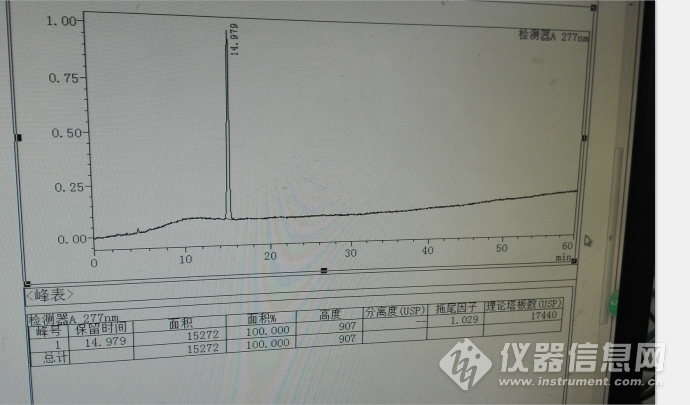
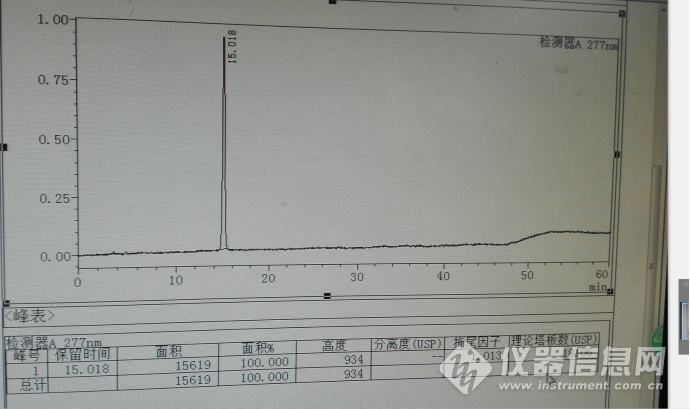
拉米夫定对照品定位溶液主峰典型色谱图如下:
![]()
![]()
以上拉米夫定对照品出峰保留时间基本上变化不大,但是浓度较大是有较大差异,15分钟在14.4分钟变化。其变化情况见下面系统色谱图。但是其相对于水杨酸的变化幅度较小。
系统进针另外图谱:
![]()
![]()
总结:我们没有用到中国药典的色谱柱,但是从色谱图及其系统适用性试验结果,其系统各杂质均能有效分离,和EP8.0提到的主峰保留时间差距很大,约6分钟差异,估计是色谱柱填料性能差异引起,从研究角度来讲,
中国产的色谱柱完全适用于本品的有关物质检测和含量测定,为我们后续的研究增强了自信心。作为权威的中国药典,不知道什么原因把色谱柱的品牌给定下来了,个人愚见,除非经大量试验比较市面主流厂家的色谱柱均不能达到要求,最好不要把色谱柱的品牌定下来。作为国产优良色谱柱的生产商月旭公司不得不由衷给个赞!