The Multi–Electrode Array DetectorThe success of the electrode array as an
$$lc detector is probably due to the development of the porous carbon electrode. This electrode is made of porous graphitic carbon, which has a very high surface area, is mechanically robust and, more important, is permeable to the mobile phase. As a consequence, flow through electrodes can be constructed. The material ideal for electrochemical detection in a number of ways. As the surface area is greatly in excess of that required for efficient electrochemical reaction, it can be severely contaminated before it fails to function. In fact, as much as 95% of the surface can be contaminated before it requires cleaning. When the electrode becomes sufficiently contaminated to require cleaning (which, according to the manufacturers, may occur between one and three years of continual use), the contamination can be rapidly removed by flushing with nitric acid.
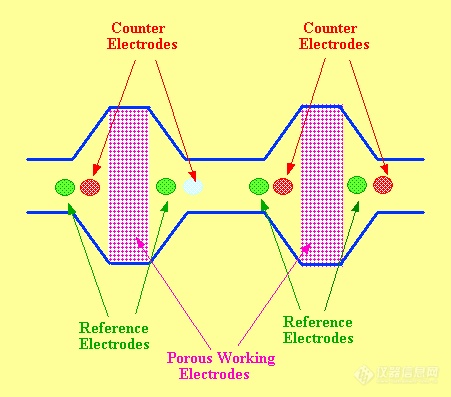
The porous graphitic carbon electrode facilitates the construction of electrode arrays. In use, the large surface area of the porous electrode ensures that 100% of the eluted material is reacted. Thus, the electrochemical reaction is no longer amperometric, but now coulometric. This is an important difference and makes the array system practical. The electrode system is shown diagramatically in figure 60.
![]() Figure 60. The Coulometric Electrode System Employing Porous Graphitic Carbon Electrodes
Figure 60. The Coulometric Electrode System Employing Porous Graphitic Carbon Electrodes