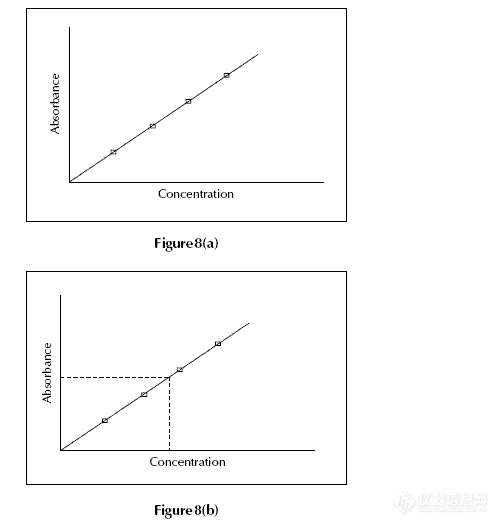
Calibration
A calibration curve is used to determine the unknown
concentration of an element – eg lead – in a solution.
The instrument is calibrated using several
solutions of known concentrations. A calibration
curve is produced which is continually rescaled as
more concentrated solutions are used – the more
concentrated solutions absorb more radiation up to a
certain absorbance. The calibration curve shows the
concentration against the amount of radiation
absorbed (Fig. 8(a)).
The sample solution is fed into the instrument
and the unknown concentration of the element – eg
lead – is then displayed on the calibration curve
(Fig. 8(b)).
![]()