7. Linearity and Detection Limits
The next few parts of this guide will provide practical information for the operator in the realization and demonstration of the key performance characteristics designed into their ICP by the manufacturer.
I have personally had the opportunity to see the advancement of ICP instrumentation over the past 30 years. Current instrumentation and software provided by manufacturers has gone well beyond anything I could have imagined 30 years ago (and it costs less). In 2004 it takes $3.21 to purchase what cost $1.00 in 1975, yet during this time period the price of ICP instrumentation has gone down while the performance characteristics have improved by orders of magnitude. A 憄ersonal experience?example is the detection limit for K by ICP-OES that has gone from 0.4 ppm to 1 ppb where the 0.4 ppm detection limit was measured on an instrument that in 1975 cost 1.5 times the 2000 price of the instrument that measured the 1 ppb detection limit (taking inflation into account, the 2000 price would be one fifth the 1975 price).
Although an ICP instrument can be wheeled into a laboratory and begin collecting data the same day, the operator is encouraged to realize and demonstrate the key performance characteristics of linearity, detectability, and spectral integrity and then go on to make decisions based upon the boundaries of these performance characteristics and the limitations of the analytical problem. Defining and being able to realize your instrument抯 performance characteristic is an investment that will save time over the years to come and allow you to make the right choices.
Defining ICP Performance Characteristics
The following steps are intended as a practical guide for the determination of an ICP抯 performance characteristics:
Read the operating manual and familiarize yourself with the software, key instrumental parameters and preferred settings before the instrument is installed.
Most instruments are supplied with optimization and wavelength or mass calibration standards that will be used during set-up by the service technician and are intended for use on a regular basis by the operator. Discuss the optimization process with the manufacturer as well as the preferred settings for the key instrumental parameters.
The remaining steps assume that the operator fully understands and is able to perform the optimization process that has been defined by the manufacturer as well as the spectral limitations of the instrument.
Select the lines to be studied for each element (憀ines?is used in this document to mean either wavelength or mass).
Line selection is based upon spectral interference issues, detection limit requirements and working range requirements. Select as many lines as possible within practicality for each element. The greater the number of lines, the greater the flexibility.
Prepare single element standards over the anticipated working range for each element. The range of standards depends upon the analytical requirements. The following ranges are suggestions only:
• Radial view ICP-OES: 0.0, 1, 10, 100, and 1000 礸/mL
• Axial view ICP-OES: 0.0, 0.1, 1, 10, and 100 礸/mL
• Quadrupole (R~ 300) mass filtered
ICP-MS: 0, 1, 10, 100, and 1000 ng/mL
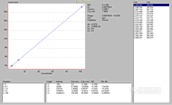
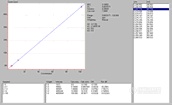
This step is important because these data can be used to determine instrument detection limits (IDL), linear working ranges, and spectral characteristics such as background equivalent concentrations (BEC) and spectral interferences. With most modern (if not all) instruments, the spectra obtained for each element at each concentration can be saved for review later. In addition, the software will calculate the IDL and BEC plus the linear regression of each line will establish the linear working range. All of this is typically done for the operator by the software that comes with the instrument. If at all possible, attempt to:
Use single element standards that have the trace metals impurities reported on the certificate of analysis. Most chemical standards manufacturers provide this information with their single element standards. These data are important in identifying direct spectral overlap interferences and in not identifying an impurity as an interference of this type.
Store all spectra on computer and collect the spectra for all lines of interest on each and every solution. This means that if you are interested in possibly using up to 6 lines for roughly 72 elements, then each solution spectrum totaling 72 x 6 = ~ 432 lines per solution and ~ 432 x 5 = 2160 spectra for each element need to be stored for future reference. Most
ICP-MS applications would require far fewer data to be collected due to the reduced number of lines available and/or feasible.
Wash blank acid solution through the instrument for several minutes 慴etween elements?and always analyze a blank at the beginning of each element concentration series. Look for the presence of the prior element analyzed to confirm that it has been completely washed out of the introduction system.
Having the data available on a desktop computer is convenient and allows the analyst to construct potential spectra by calling up the element and the anticipated concentration for each element in the analytical sample. Having several lines available makes the job of line selection easy as well as the estimation of the line抯 sensitivity and linearity. Constructing these composite spectra from pure single element solutions eliminates confusion as to the identity of the line. The following example is intended to illustrate the process:
Examples of Spectra:
NOTE: All spectra were obtained using a concentric glass nebulizer with no problems around salting out or plugging.
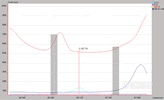
The following example is for an application where a submitter has been obtaining minor levels (0.1 to 1.0 %) of Cr in an alloy containing roughly equal amounts of Fe and Ni. The laboratory where this alloy is analyzed uses a procedure where 0.2 grams of the sample is dissolved in 5 mL of a 1:1 HNO3 / HCl mixture and diluted to 1000 mL with DI water. The analyst is informed that a limit of detection (LOD = 3SD0) of 1 ppm Cr based upon the original sample and the ability to quantify the Cr to within &plmn;10 % relative at the 10 ppm level is an absolute minimum requirement.
The submitter then asks the analyst the usual question, 揑 need the results tomorrow ?can you do it??The analyst does a quick calculation and determines that using the most sensitive Cr line and the current procedure, the lowest possible detection limit is 4 ppm and a more realistic estimation would be ~ 4 times the IDL or ~ 16 ppm. The analyst then pulls up the following spectra, instrument detection limits, and linear regression data which were obtained on their radial view instrument about four years ago when installed using pure single element solutions as described above.