
原文由 happyjyl 发表:
下面这份注册申请表是翻译给国外供应商看的,放上来大家讨论。涉及到专业方面的内容很少,主要是行政方面的,大家都能看明白的。有些地方我觉得老外可能不明白,就自己加了注释。
表头:
数据核对码:Bar Code[1]
原始编号:Original Application No. [2]
受理号:Accept No. [3]
国家食品药品监督管理局
STATE FOOD AND DRUG ADMINISTRATION
药品注册申请-境外申请人用表
DRUG REGISTRATION APPLICATION -- FOR FOREIGN APPLICANTS
[1]: The bar code is created randomly by the software. Each time the application form is amended, a new bar code will be created.
[2], [3]: To be given by SFDA after the application is accepted.
原文由 redanqi 发表:原文由 happyjyl 发表:
[1]: The bar code is created randomly by the software. Each time the application form is amended, a new bar code will be created.
[2], [3]: To be given by SFDA after the application is accepted.
受理号和原始编号有什么区别?
原文由 cbjcn1985 发表:原文由 happyjyl 发表:
药品情况
11商品名:无
Brand Name: none
12制剂类型:非制剂 原料药
Product category: API
13规格:
Strength:
11.Trade name
12.formulation type
13.specification
这几点,这样翻译可以嘛??只是个人的看法...犯错误,请指正
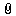 进口药品注册申请表
进口药品注册申请表